Nomenclature
1. Introduction
2. Results and Discussions
2.1 Synthesis and characterizations of COE ligands
2.3 Relationship between FRET efficiency and ionic density of COE ligands
3. Conclusions
4. Experimental Details
4.1 Materials
4.2 General
4.3 Synthesis of TFs
4.4 Synthesis of COEs
4.5 Synthesis of Colloidal PNCs
Nomenclature
Metal-halide perovskite nanocrystal : PNC
Photoluminescence quantum yield : PLQY
Oleyl amine & oleic acid : OA
Conjugated oligoelectrolyte : COE
Förster resonance energy transfer : FRET
UV photoelectron spectroscopy : UPS
Charge-neutral terfluorene precursor : TF
Trifluoroethanol : TFE
Nuclear magnetic resonance : NMR
Transmission electron microscopy : TEM
PL lifetime : τ
1. Introduction
Metal-halide perovskite nanocrystals (PNCs) have gained an interest as light-emissive materials due to their high photoluminescence quantum yield (PLQY), narrow emission bandwidth, and facile color tunability through managing halide composition1,2,3,4). A crucial determinant of their performance lies in the surface ligand, which controls colloidal stability, defect passivation, and interfacial electronic coupling. Conventional long-chain aliphatic ligands (e. g. combination of oleyl amine & oleic acid (OA)) are adopted for PNC ligand for their stabilizing ability in colloidal PNCs. However, PNCs synthesized with OA usually exhibit poor passivation due to their loosely bound nature and insufficient surface halides requiring additional treatment5). In addition, OA forms insulating shells that usually hinder the optoelectronic performances in their actual application6, 7). To overcome these limitations, conjugated ligands with strong binding nature which can also facilitate charge delocalization at the PNC surface by π-systems have been adopted8).
Meanwhile, we have developed a conjugated oligoelectrolyte (COE), consisting of a terfluorene-based oligomeric backbone functionalized with two quaternary ammonium bromides for the ligand of CsPbBr3 PNCs9). This ligand not only binds strongly to the PNC surface but also enables efficient Förster resonance energy transfer (FRET) from the COE to the PNCs in the colloidal state, achieving a FRET efficiency of 43%. A key factor in COE design is the incorporation of multiple ionic anchoring groups within each COE molecule. Therefore, higher ionic density in COE is expected to increase binding intimacy between the ligand and PNC surface, leading to more effective passivation and stronger dipole-induced modulation of surface energetics.
Herein, we introduce a blue-fluorescent COE ligand based on a fluorene trimer backbone, functionalized with quaternary ammonium halides. This design allows simultaneous optical and electronic functionality: (i) the extended π-conjugation facilitates charge delocalization and blue emission and (ii) increased number of ionic side chains from 2 to 4 and 6, enhancing the binding intimacy with CsPbBr3 PNCs to enhance FRET efficiency. By systematically varying the ionic density of the COE, we correlate ligand-PNC intimacy with FRET efficiency, thereby providing direct insights into how molecular design dictates interfacial coupling.
2. Results and Discussions
2.1 Synthesis and characterizations of COE ligands
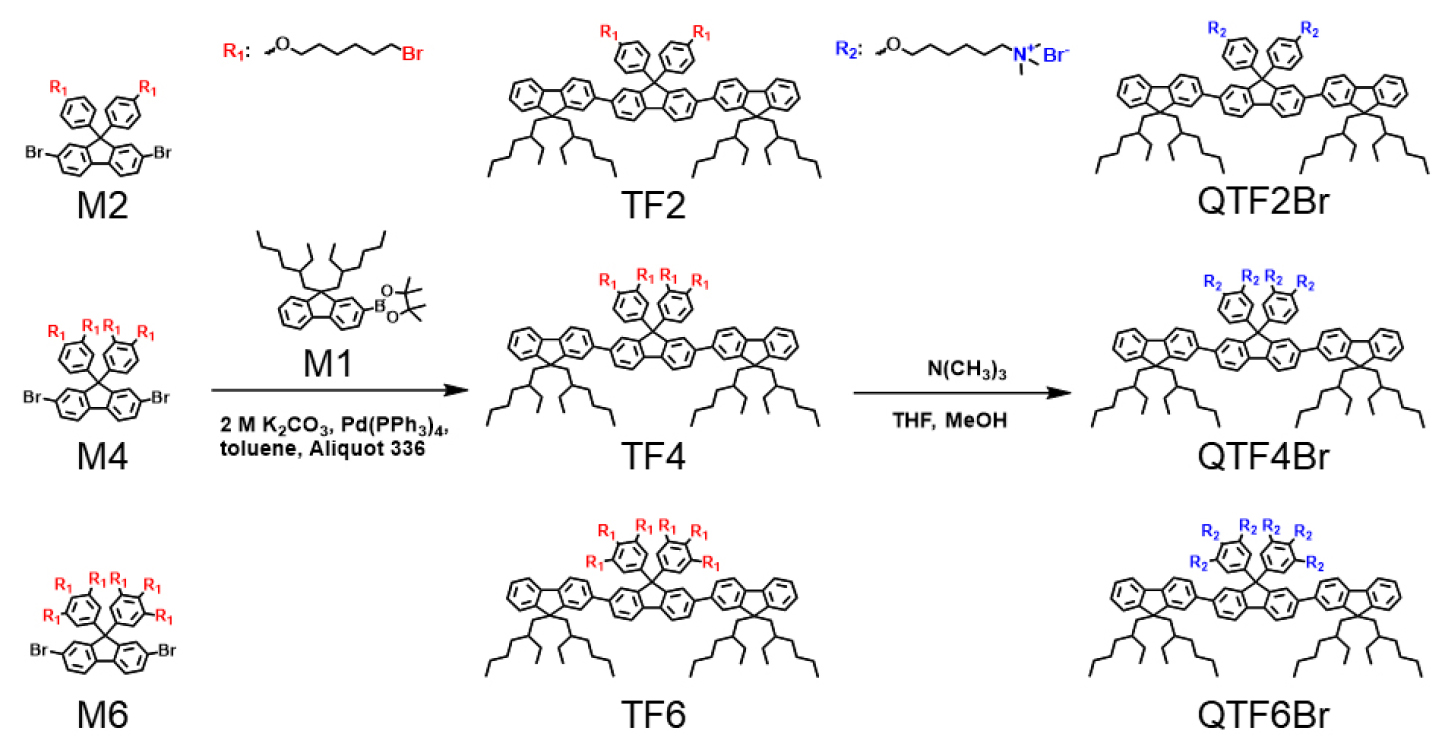
Fig. 1 summarizes the synthetic routes and chemical structures of the COEs which is modified from the reported synthetic procedure of QTF2Br9). The neutral terfluorene precursors (TF4 and TF6) were primarily synthesized by stepwise Suzuki-Miyaura coupling of bromofluorene and two fluorene boronate intermediates, producing the terfluorenes with multiple bromoalkyl terminals. These charge-neutral terfluorene precursors (TFs), namely TF2, TF4, and TF6 were subsequently quaternized by reaction with excess trimethylamine under room temperature, affording three target COEs with 2-, 4- and 6- terminal trimethyl ammonium bromides functionalities.
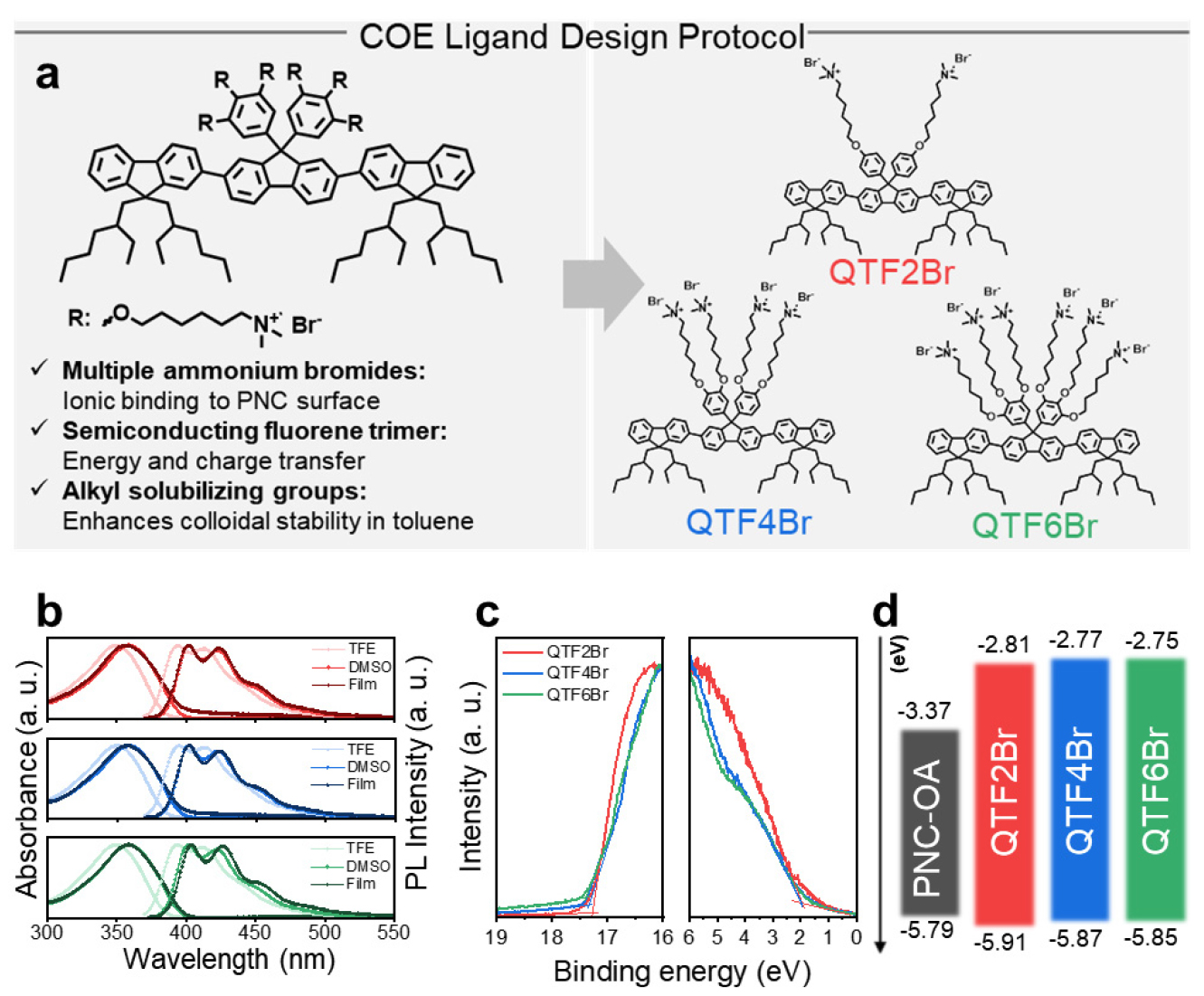
Fig. 2 (a) describes the molecular design protocol and role of each segment in COE. The synthesized COEs with terfluorene- based conjugated core exhibit strong π-π* absorption bands centered at ~360 nm, with a relatively large optical bandgap (~3.1 eV) and blue fluorescence (λPL ≈ 418 nm), typical of fluorene oligomers (Fig. 2 (b))10, 11). Notably, the emission spectrum of the COEs partially overlaps with the broad absorption band of CsPbBr3 PNCs, creating favorable conditions for FRET from COE donors to PNC acceptors. This spectral overlap, combined with the short donor-acceptor separation achievable via ionic binding, motivated us to investigate the COEs as multifunctional ligands for PNCs. According to the energy level diagram of COEs and PNC obtained from UV photoelectron spectroscopy (UPS), the energy band of PNC lies between those of COEs which act an effective role for FRET with suppressed back transfer of hole from PNC to COE (Fig. 2 (c), (d)).
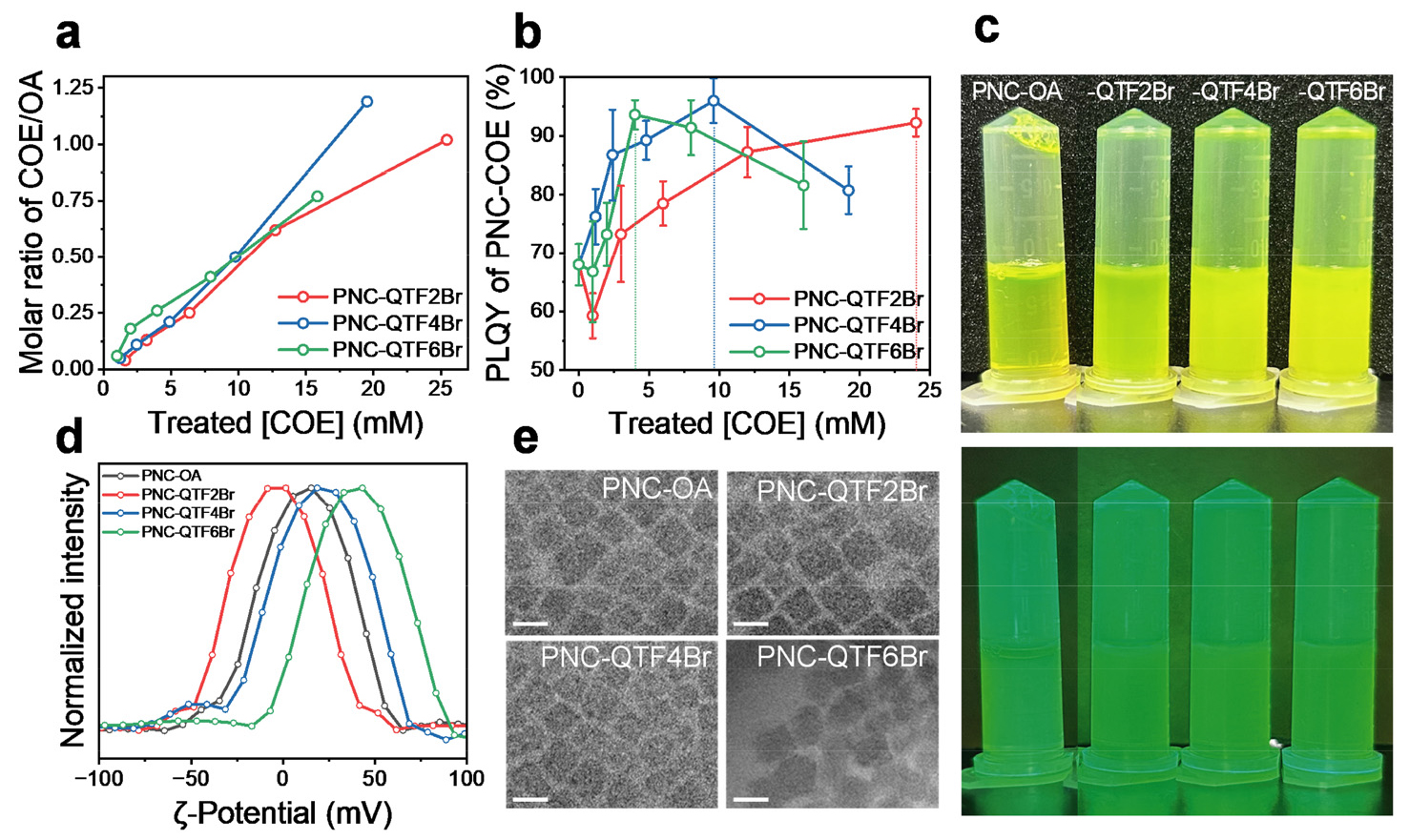
Colloidal CsPbBr3 PNCs with the native ligands of OA capping ligands (PNC-OA) and PNC-COEs, namely, PNC-QTF2Br, -QTF4Br, and -QTF6Br were prepared based on the modified synthetic protocol of QTF2Br (see Experimental Section for details). To determine the optimal ligand exchange conditions depending on the type of COEs, we evaluated the absolute PLQY trends with varying concentration of treated COE in trifluoroethanol (TFE) solution (2.5, 5.0, 10, 20, and 40 mg mL−1). Higher treatment of [COE] is anticipated to yield better PNC quality because not only can the adequate amount of COEs counteract the stripping of OAs, but also COE can provide extra halide to bind with exposed Pb at PNC surface. The ligand molar ratio of COE/OA was first identified by nuclear magnetic resonance (NMR) (Fig. 3 (a)). During ligand substitution with COE, the native OA ligands were stripped off by TFE, allowing COEs to occupy the vacant sites12, 13). According to NMR, increasing the treatment concentration of COE led to a nearly linear increase in the molar ratio of COE incorporated in the resulting PNC-COEs. To evaluate the quality of the PNCs, we further monitored the absolute PLQY variations as a function of COE treatment concentration.
Interestingly, PNC-QTF2Br, -QTF4Br, and -QTF6Br showed different dispersibility and PLQY trends. In our previous report, optimal [QTF2Br] for the synthesis of PNC-QTF2Br is 40 mg mL−1 (= 25.5 mM) with PLQY of 94%. In the case of QTF4Br, reached highest PLQY of 96% at 20 mg mL−1 (= 9.8 mM), indicating more efficient halide-rich passivation by doubled Br− supply from QTF4Br. However, at 40 mg mL−1 (= 19.5 mM) of [QTF4Br], dispersibility and PLQY decreased, producing turbid PNC solution (Fig. 3 (b), (c)). This trend became even more pronounced in the case of PNC-QTF6Br, where the highest PLQY (93.6%) was achieved at only 10 mg mL−1 (= 4.0 mM). Beyond this concentration, a gradual decrease in PLQY was observed. Consequently, the highest absolute PLQY was shown in 40 mg mL−1-, 20 mg mL−1-, and 10 mg mL−1- treated COE solutions for PNC-QTF2Br, PNC-QTF4Br, and PNC-QTF6Br, being the optimal ligand substitution condition for the PNC-COEs, respectively. ζ-potential measurements were conducted to assess the surface charge states and corresponding dispersibility of the colloidal state resulting from optimal ligand exchange condition (Fig. 3 (d)). For PNC-OA, the ζ-potential was at +12.3 mV which is a near-zero potential that can stabilize the surface in non-polar media such as toluene. While the ζ-potential of PNC-QTF2Br showed at +1.54 mV, PNC-QTF4Br, and -QTF6Br at their optimal PLQY exhibited ζ-potentials of +18.6, and +43.1 mV, respectively, indicating the formation of more polar surface on the shell14). This result is likely due to the presence of surface-oriented cationic ammonium groups from the multifunctional COEs, which hinders the dispersibility of PNCs in toluene. According to the transmission electron microscopy (TEM) images of PNCs, the pre-synthesized PNC-OA exhibited cubic-shaped nanocrystals with a size of 10 nm (Fig. 3 (e)). Similarly, both PNC-QTF2Br and PNC- QTF4Br treated with 40 and 20 mg mL−1 COE solutions, respectively, retained the same cubic morphology, indicating no observable surface damage or deformation caused by ligand exchange. In contrast, PNC-QTF6Br treated with 10 mg mL−1 displayed a rounding of the crystal edges, which is likely attributed to the promotion of binding with facets other than the [100] planes, induced by the random arrangement of the multicharged ionic density.
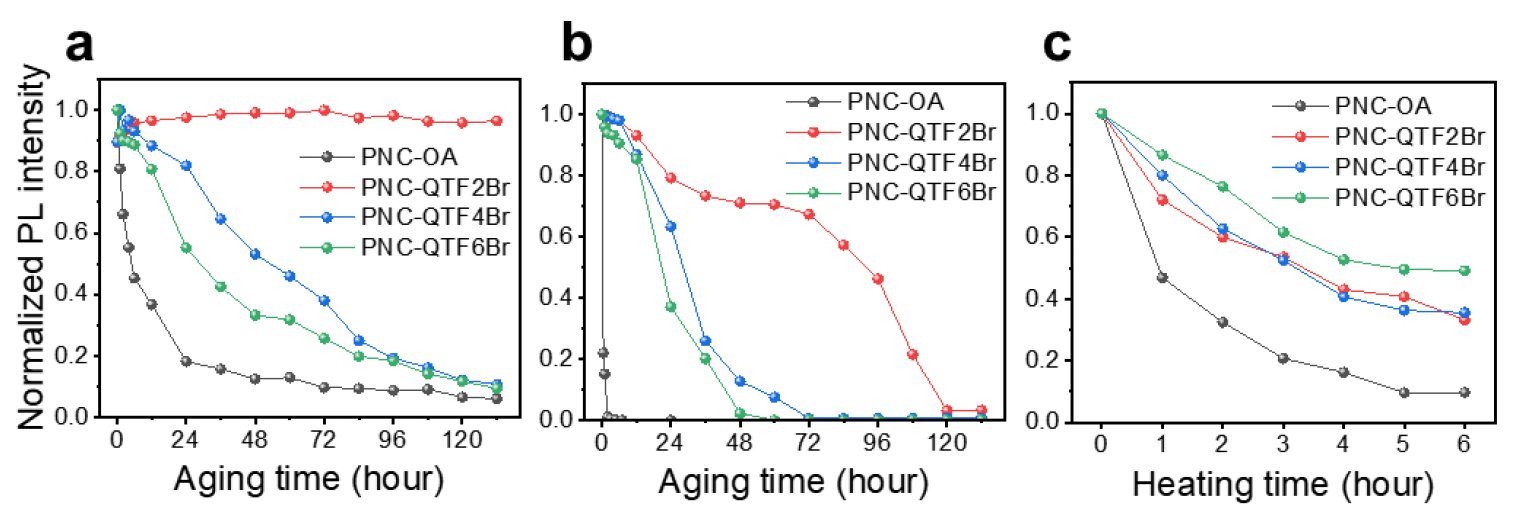
To assess the colloidal and thermal stability of the PNCs, the PL evolution of respective colloidal dispersions in toluene and tetrahydrofuran (THF) was monitored under ambient conditions (RH ≈ 80%) over 132 hours (Fig. 4 (a), (b)). While PNC-OA rapidly lost most of its photoluminescence within 24 hours, all PNC-COE samples exhibited markedly enhanced stability, with PNC-QTF2Br maintaining nearly constant PL intensity throughout the test period. In THF, the difference became even more evident―PNC-QTF2Br, QTF4Br, and QTF6Br required approximately 120, 60, and 48 hours, respectively, to degrade below 10% of their initial PL, whereas PNC-OA degraded completely within one hour. Furthermore, PL tracking of the corresponding thin films (spin-coated on glass substrate at 1500 rpm) heated at 100°C under ambient atmosphere revealed significantly improved thermal robustness for PNC-COE compared to PNC-OA (Fig. 4 (c)). In particular, PNC-QTF6Br retained more than 50% of its initial PL after 6 hours of heating, suggesting that stronger ionic binding at higher ionic densities contributes to enhanced solid-state stability. These observations support that ionic conjugated ligands play a critical role in improving both colloidal and thermal stability of PNCs.
2.3 Relationship between FRET efficiency and ionic density of COE ligands
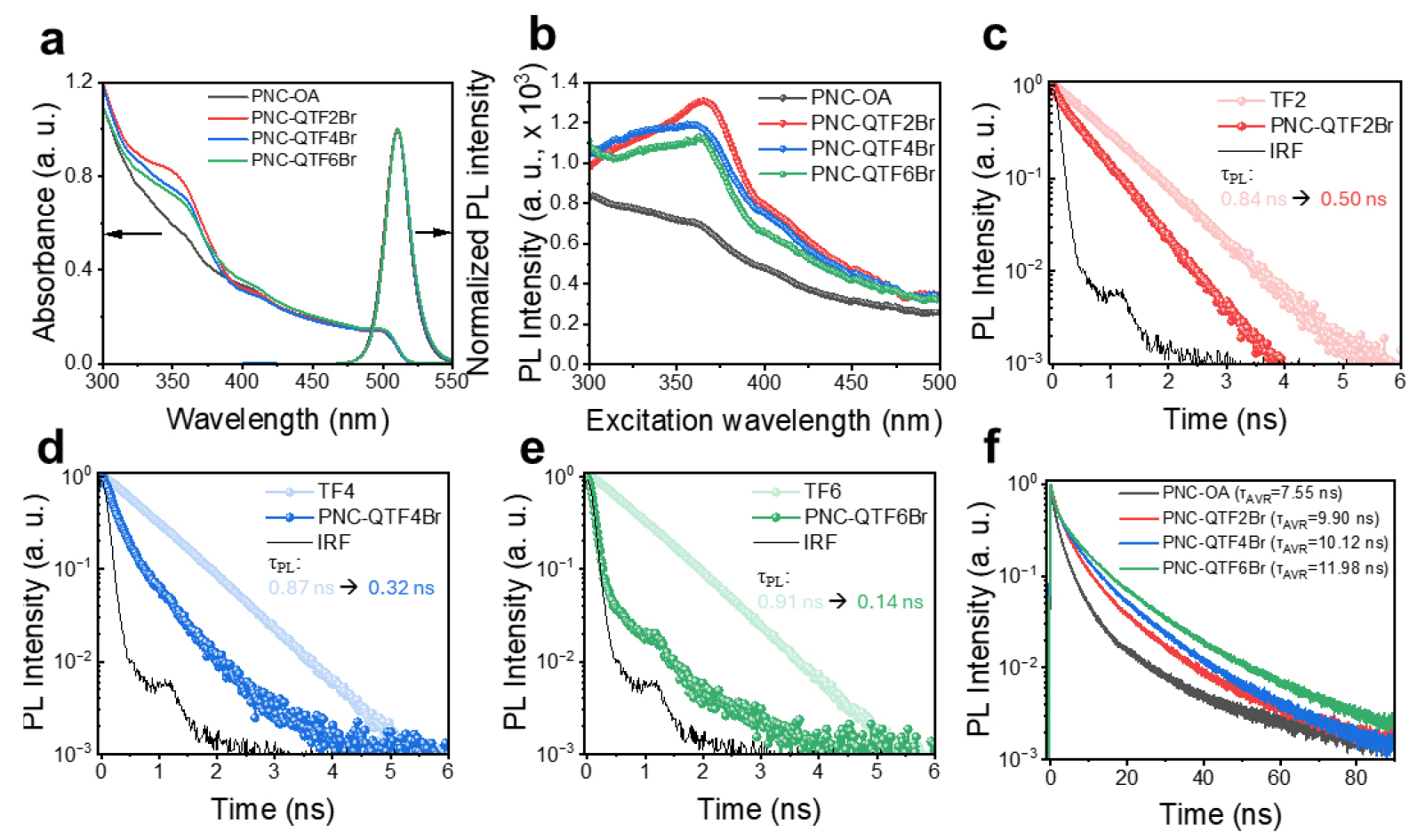
Optical characterization provides further evidence of ligand incorporation and reveals the FRET behavior in PNC-COEs. After ligand exchange, the UV-vis spectra exhibited an absorption peak at around 360 nm in PNC-COEs, corresponding to the π-π* transition of the terfluorene unit, thereby confirming the presence of COEs on the PNC surface (Fig. 5 (a)). The absorbance arising from COEs varied among different ligands, reflecting differences in their molar content. According to the PL spectra in Fig. 5 (a), the narrow emission peak of CsPbBr3 at 513 nm remained unchanged after COE substitution, indicating that the ligand exchange did not induce degradation of the nanocrystals. Furthermore, no intrinsic blue emission from the COEs was observed in the PNC-COE samples, despite clear COE absorption. Photoluminescence excitation (PLE) spectra monitored at 513 nm emission are shown in Fig. 5 (b). For PNC-OA, the PL intensity as a function of excitation wavelength followed the optical density trend of the UV-vis absorbance spectrum. In contrast, PNC-COEs exhibited enhanced PL intensity at 513 nm when excited in the 300-400 nm region, where COE absorption occurs. The absence of distinct fluorescence from the COEs, combined with the direct contribution of COE absorption to the 513 nm emission of PNCs, further supports the occurrence of FRET in PNC-COEs.
Time-resolved PL (TRPL) measurements at the COE emission wavelength (418 nm) provided quantitative insight into the FRET dynamics. While the COEs themselves possess highly polar characteristics and are insoluble in toluene, TFs (which have same terfluorene backbones with respective COEs but different termina) exhibit good solubility in toluene. Therefore, their solutions can be regarded as being in a completely free ligand state without any FRET. TRPL measurements of respective TFs revealed mono-exponential decays with PL lifetimes (τ(TF)) of ~0.8-0.9 ns (Fig. 5 (c)-(e)). In the case of PNC-COEs, the COE donor lifetimes were progressively shortened to 0.50 ns (QTF2Br), 0.32 ns (QTF4Br), and 0.14 ns (QTF6Br), clearly indicating FRET from COE to PNC. These accelerated decays directly reflect efficient non-radiative energy transfer, with calculated FRET efficiencies (=1 − τ(PNC-COE)/τ(TF)) of 40%, 63%, and 84%, respectively15). The monotonic increase in efficiency with ionic density highlights the role of strong electrostatic binding in bringing donor and acceptor into close proximity, thereby enhancing dipole-dipole coupling. Meanwhile, at the PNC emission wavelength of 513 nm, the TRPL profiles exhibited bi-exponential decay components. PNC-OA showed an average PL lifetime of 7.55 ns, whereas PNC-QTF2Br, PNC-QTF4Br, and PNC-QTF6Br exhibited progressively increasing average PL lifetimes of 9.90, 10.12, and 11.98 ns, respectively (Fig. 5 (f)). This extension of τAVR indicates effective suppression of non-radiative pathways through defect reduction, which becomes more pronounced with increasing ionic density16).
3. Conclusions
This study demonstrates that the ionic density of the COE ligands is a key determinant of both colloidal stability and FRET efficiency for PNC-COEs to improve their PL properties. Increasing the ionic density of COEs leads to the highest PLQY of the corresponding PNC-COEs at lower COE content, owing to the higher ion density and more effective surface passivation. However, this benefit comes at the expense of reduced PNC dispersibility, resulting from the increased ionic content on the PNC surface. A moderate ionic density of QTF2Br balances good dispersibility and substantial FRET, while higher ionic densities of QTF4Br and QTF6Br show more efficient energy transfer owing to their intimate binding to CsPbBr3 surface. This systematic variation provides direct molecular-level insight into how ionic functionalities modulate binding intimacy and photophysical coupling at the PNC surface, informing the rational design of multifunctional semiconducting ligands for perovskite nanocrystals. Prospectively, this trade-off can be addressed by controlling the mixing ratio of additional ligands, which could provide a balanced optimization between surface passivation and colloidal stability. Beyond enhancing PLQY and FRET efficiency, ionic-density engineering in COE structures can facilitate the development of stable, high-performance perovskite fluorescent probes and extend their use to light-emitting devices, solar cells, and other practical optoelectronic applications. This paves the way for integrating perovskite nanocrystals into next-generation optoelectronic technologies.
4. Experimental Details
4.1 Materials
All the reagents for the synthesis of COEs and PNCs are purchased from Sigma Aldrich and Tokyo Chemical Industry.
4.2 General
1H and 13C NMR spectra were recorded on a Bruker Advance III HD system operating at 500 MHz and 125 MHz, respectively. TEM images of perovskite nanocrystals were obtained using a JEOL JEM-2010 microscope. UV-Vis absorption spectra were measured with a Jasco V-770 spectrometer. Steady-state PL and PLE spectra of colloidal perovskite nanocrystals were collected using a Jasco FP-8600 spectrofluorometer with spectrum correction applied. ζ-Potential measurements were conducted on an Otsuka ELSZ-1000 instrument, applying the Hückel approximation for non-polar systems. UPS was performed using a Thermo Fisher Scientific Nexsa system with an Al Kα monochromatic source.
4.3 Synthesis of TFs
M1, M2, M4, M6, and TF2 were synthesized via reported synthetic methods9, 17, 18). The neutral trimers were synthesized by Suzuki coupling. For the synthesis of TF4 or TF6, 0.60 mmol of M4 (0.71 g) or M6 (0.93 g) was respectively loaded with M1 (1.26 mmol, 0.65 g), Pd (PPh3)4 (6 mmol%, 0.04 g), and aliquot 336 (2 drops) in a 20 mL microwave vial. The mixture was dissolved in anhydrous toluene (4 mL), and 2 M of aqueous K2CO3 solution (2 mL) was added. The reaction mixture was stirred at 90 °C for 24 h. The crude solution was extracted with ethyl acetate, dried over MgSO4, and filtered. After evaporating solvent, the crude product was purified by column chromatography using hexane and ethyl acetate as an eluent. (hexane/ethyl acetate = 50:1 v/v). After the solvent was totally removed under the vacuum, colorless viscous products of TF4 and TF6.
TF4: Yield: 0.44 g (41%); 1H-NMR (500 MHz, CDCl3): δ (ppm) 7.86-7.84 (m, 2H), 7.71-7.70 (m, 4H), 7.69-7.63 (m, 4H), 7.56 (d, 2H), 7.50-7.40 (m, 2H), 7.39-7.36 (m, 2H), 7.34-7.30 (m, 2H), 7.28-7.25 (m, 2H), 6.91-6.86 (m, 2H), 6.84-6.75 (m, 2H), 6.73-7.69 (q, 2H), 3.95-3.90 (m, 4H), 3.85-3.82 (m, 4H), 3.55-3.33 (m, 8H), 2.05-1.97 (m, 8H), 1.89-1.72 (m, 16H), 1.50-1.40 (m, 16H), 0.94-0.63 (m, 40H), 0.56-0.46 (m, 20H).
TF6: Yield: 0.66 g (51%); 1H-NMR (500 MHz, CDCl3): δ (ppm) 7.88-7.85 (m, 2H), 7.75-7.68 (d, 4H), 7.68-7.65 (m, 4H), 7.57-7.55 (s, 2H), 7.51-7.48 (d, 2H), 7.40-7.27 (m, 6H), 6.51-6.45 (s, 4H), 3.91-3.85 (m, 4H), 3.78-3.70 (m, 8H), 3.52-3.35 (m, 12H), 2.50-1.81 (m, 8H), 1.91-1.64 (m, 24H), 1.54-1.42 (m, 24H), 0.93-0.64 (m, 40H), 0.54-0.44 (m, 20H).
4.4 Synthesis of COEs
Synthesis of QTF2Br, QTF4Br, and QTF6Br: Multiply charged COEs were synthesized by following quaternization protocols of QTF2Br in our previous report9). In a 1-neck round bottom flask, TF4 or TF6 (0.12 mmol) was dissolved in THF (6 mL). 3.2 M trimethylamine solution in methanol (2.4 mmol, 0.75 mL) was added dropwise slowly in the reaction mixture. The reaction mixture was stirred at room temperature for 24 h. During the reaction, the additional amount of methanol (1 mL) was added in the reaction mixture to put precipitated intermediates in further reaction. After removing the solvent in vacuo, the reaction was repeated for total conversion. Consequently, crude mixture was precipitated in diethyl ether, filtered, and dried in vacuo sequentially to obtain the respective COEs.
QTF4Br: Yield: 0.15 g (79%); 1H-NMR (500 MHz, DMSO-d6): δ (ppm) 8.10-8.08 (m, 2H), 7.88-7.83 (m, 4H), 7.81-7.75 (m, 2H), 7.73-7.69 (m, 4H), 7.62-7.60 (q, 2H), 7.51-7.49 (m, 2H), 7.36-7.29 (m, 4H), 6.91-6.73 (m, 6H), 3.90-3.74 (m, 4H), 3.76-3.74 (m, 4H), 3.32-3.20 (m, 8H), 3.09-2.98 (m, 36H), 2.12-1.97 (m, 8H), 1.70-1.60 (m, 16H), 1.45-1.34 (m, 8H), 1.32-1.19 (m, 8H), 0.88-0.55 (m, 40H), 0.48-0.38 (m, 20H).
QTF6Br: Yield: 0.27 g (90%). 1H-NMR (500 MHz, DMSO-d6): δ (ppm) 8.13-8.07 (m, 2H), 7.92-7.71 (m, 14H), 7.70-7.62 (m, 2H), 7.51-7.50 (m, 2H), 7.37-7.30 (m, 4H), 6.48-6.46 (s, 4H), 3.78-3.73 (m, 12H), 3.27-3.24 (m, 12H), 3.04-3.02 (s, 54H), 2.05-2.03 (m, 8H), 1.66-1.59 (s, 24H), 1.41-1.36 (m, 12H), 1.27-1.22 (s, 12H), 0.86-0.53 (m, 40H), 0.49-0.31 (m, 20H).
4.5 Synthesis of Colloidal PNCs
For the synthesis of CsPbBr3 PNCs, PNC-OA readily synthesized following the reported hot-injection protocol19). Inside an N₂-filled glove box, the as-synthesized PNC-OA solution (10~12 mg/mL, 200 μL in toluene) was mixed with 20 μL of a COE in TFE (concentration range: 2.5-40 mg/mL) and vortexed for 3 minutes. Subsequently, 400 μL of MeOAc was put into the solution and the mixture was gently agitated until a cloudy suspension appeared, followed by centrifugation at 13,500 rpm for 3 minutes. After discarding the supernatant, the resulting precipitate was redispersed in 200 μL of toluene to afford the PNC-COE stock solutions. The stock solution was diluted 300-fold in toluene and used for optical measurements.