Subscript
1. Introduction
2. Experimental Details
2.1 Synthesis of BiVO4 photoanode
2.2 Analysis methods
3. Results and Discussion
4. Conclusions
Subscript
PEC : photoelectrochemical
HER/OER : hydrogen/oxygen evolution reaction
BiVO4 : bismuth vanadate
KPi : potassium phosphate
Na2SO4 : sodium sulfate
CoPi : cobalt phosphate
OEC : oxygen evolution catalyst
FTO : fluorine-doped tin oxide
PED : (Photo-) electrodeposition
CA : chronoamperometry
KB : potassium borate
LSV : linear sweep voltammetry
ABPE : applied bias photon-to-current efficiency
EIS : electrochemical impedance spectroscopy
PCET : proton-coupled electron transfer
1. Introduction
Driven by the urgency of climate change and the limitations of fossil fuels, there is a growing emphasis on developing clean and renewable energy technologies such as solar energy, green hydrogen and carbon reduction. Photoelectrochemical (PEC) water splitting presents a compelling approach for sustainable hydrogen generation due to its potential for direct solar-to-fuel conversion and environmental compatibility1-3). PEC water splitting offers the distinct advantage of directly generating hydrogen from water under solar illumination, independent of external conditions, and without producing harmful byproducts, thereby enabling the production of truly green hydrogen. This technology has demonstrated competitiveness in terms of high energy conversion efficiency, a wide range of material choices, and cost-effective processing, prompting active research efforts toward commercialization4). However, several technological limitations remain, particularly concerning key components such as oxide semiconductor-based photoabsorbers, conductive substrates, overlayers, catalyst, and electrolytes, which hinder improvements in both PEC efficiency and long-term stability5-8).
Most previous studies have focused primarily on developing hydrogen evolution reaction (HER) systems based on various p-type oxide semiconductors, including Cu2O, CuO, NiO, Co3O4, CuFeO2, and CuBi2O4 as photocathode materials9-15). In contrast, the oxygen evolution reaction (OER), which is essential for complete water splitting, involves a complex four-electron transfer process, and is inherently accompanied by high overpotentials and poor stability. In this context, n-type semiconductor materials such as BiVO4, Fe2O3, WO3, TiO2, TaON, SrTiO3, and g-C3N4 are typically employed as photoanodes. Therefore, the development of efficient photoanodes capable of driving OER is critically important16). In particular, to realize self-driven PEC systems, it is essential to suppress the OER overpotential by designing high-performance photoanodes. Such electrodes are key to reducing the overall energy input required by the system and enabling water splitting without the need for an external bias, thereby functioning as decisive components in self-sustained solar hydrogen production. Among the various n-type oxide semiconductors investigated for photoanode development, bismuth vanadate (BiVO4) has emerged as one of the most extensively studied materials because of its structural stability, suitable bandgap (2.4 eV), and relatively high theoretical solar-to-hydrogen efficiency of approximately 9.2%17-20). In particular, BiVO4 photoanodes exhibit excellent visible light absorption but suffer from intrinsic limitations such as severe bulk and surface charge recombination and low charge carrier mobility, which necessitates further optimization to realize high-performance PEC devices21). As minority carriers (holes) must reach the photoanode surface to initiate the oxygen evolution reaction, balancing light absorption and hole transport is crucial. Given the short hole diffusion length of approximately 70–100 nm in BiVO4, excessively thin films result in insufficient light harvesting, whereas excessively thick films increase the bulk recombination losses, thereby reducing the PEC efficiency22). Although nano-structuring can reduce the carrier transport distance in thin-film BiVO4 photoanodes, strategic control over the film thickness is essential to optimize the trade-off between absorption and charge transport.
In addition, the electrolyte environment plays a critical role in determining the PEC performance and stability. From a practical standpoint, achieving stable PEC performance under near-neutral pH conditions (6–8) is highly important, particularly for deployment in systems that utilize natural water sources, including seawater and buffered solutions. Therefore, the development of additional stabilization strategies is necessary to ensure the long-term durability of photoanodes without compromising their activities in neutral or near-alkaline electrolytes. However, depending on the electrolyte used, a trade-off often exists between catalytic activity and stability. For instance, potassium phosphate (KPi) buffer provides high OER activity but insufficient long-term stability, whereas sodium sulfate (Na2SO4) offers relatively better stability at the expense of lower OER activity23-25). This electrolyte-dependent trade-off highlights the need for a co-engineered stabilization approach that can simultaneously satisfy both the activity and durability requirements in practical PEC applications.
To address this issue, we introduced a cobalt phosphate (CoPi) oxygen evolution cocatalyst (OEC) onto BiVO4 photoanodes to simultaneously enhance the OER activity and the long-term stability of the KPi electrolyte. CoPi not only maintains and improves catalytic activity but also protects the BiVO4 surface by mitigating oxidative degradation and structural deterioration, enabling high performance and extended durability in the KPi environment. Notably, the continuous supply of phosphate ions from the KPi electrolyte stabilizes the high-valent CoIV species in CoPi, reinforcing both the structural integrity and electrochemical reactivity, thereby producing a synergistic effect that enhances the PEC performance. Whereas prior studies on CoPi have primarily focused on enhancing OER activity, the present work distinguishes itself by targeting long-term stability as a co-objective. This strategy aims to overcome the intrinsic limitations of KPi-based PEC systems and realize stable and efficient water splitting operations. By applying a photo-assisted electrodeposited (PED) CoPi OEC onto a BiVO4 photoanode with an optimized thickness (~120 nm), we successfully mitigated the instability observed in pristine BiVO4 under KPi conditions and achieved a high photocurrent density of 2.8 mA/cm2, with only a 12.86% decrease after more than 10 hours of continuous PEC operation.
2. Experimental Details
2.1 Synthesis of BiVO4 photoanode
BiVO4 thin films were prepared via a solution-based sol–gel method. Initially, 50 × 10-3 M bismuth nitrate was dissolved in 20 mL of aqueous acetic acid and stirred at 350 rpm for 1 h. Subsequently, 46.5 × 10-3 M vanadyl acetylacetonate was slowly added to the solution and stirred for an additional 15 min at the same speed. To adjust the viscosity and promote film formation, 0.2 g of ethyl cellulose was added as an organic binder to complete the precursor solution. The resulting solution was spin-coated onto pretreated fluorine-doped tin oxide (FTO, 10 Ω/sq.) glass substrates at 3000–4500 rpm for 40 s. Prior to the deposition, the FTO substrates were ultrasonically cleaned in acetone, ethanol, and DI water for 20 min each to remove surface contaminants. The coated substrates were soft-baked on a hot plate at 550°C for 30 min to induce pre-crystallization, followed by air-drying at room temperature for 30 min. The coating and pre-baking steps were repeated as required to achieve the desired film thickness. Finally, the BiVO4 films were hard-baked in a tube furnace in ambient air at 550°C for 2 h at a heating rate of 2°C/min.
A CoPi cocatalyst was introduced onto the surface of the BiVO4 photoanode via a conventional electrodeposition (ED) process. The deposition electrolyte was prepared by dissolving 0.5 × 10-3 cobalt(II) nitrate hexahydrate (Co(NO3)2·6H2O), 6.5 × 10-3 M potassium phosphate dibasic (K2HPO4), and 3.4 × 10-3 M potassium phosphate monobasic (KH2PO4) in 300 mL of DI water, and the solution was buffered to pH 7.0. CoPi deposition was performed for 90 s using chronoamperometry (CA) in a buffered electrolyte solution. For comparison, the PED method was employed to evaluate the effect of photoactivity on the catalyst deposition behavior. Under the PED condition, the BiVO4 electrode was irradiated with simulated 1 sun (100 mW/cm2) through a 1.5 G optical filter while using the same electrolyte composition and CA conditions for 90 s. This approach enabled the investigation of the influence of photogenerated charge carriers on CoPi nucleation and growth, as well as the resulting impact on the PEC performance. The deposition process was conducted at an applied potential of 0.2 V versus Ag/AgCl (reference electrode saturated with 4 M KCl, VAg/AgCl). The potential versus Ag/AgCl was converted to the reversible hydrogen electrode (RHE) scale using the Nernst equation:
2.2 Analysis methods
The cross-sectional morphology of the fabricated BiVO4 photoanode was analyzed using field-emission scanning electron microscopy (FE-SEM; JSM-6700F, JEOL). The optical properties of the photoanode were characterized using a UV-Vis-NIR spectrophotometer (Cary 5000, Agilent) equipped with an integrating sphere over a wavelength range of 300–800 nm.
Photoelectrochemical (PEC) measurements were conducted using a standard 3 electrode configuration under illumination from a solar simulator (HAL-320, ASAHI SPECTRA) equipped with an AM 1.5G filter, controlled by a Versastat 3 system (AMETEK). The three-electrode setup consisted of a BiVO4-based photoanode as the working electrode, saturated Ag/AgCl electrode (with 4 M KCl) as the reference electrode, and a platinum (Pt) mesh as the counter electrode.
The electrolytes used included a 1.0 M potassium borate buffer (KB) solution, prepared by adjusting the pH of a H3BO3 solution to 9.5 using KOH, as well as 1.0 M KPi buffer solution and 1.0 M Na2SO4 solutions, both adjusted to pH 7.0. For sulfite oxidation conditions, 0.1 M Na2SO3 was additionally introduced into the respective buffered electrolytes. During measurements, photoelectrodes with an exposed area of approximately 0.25 cm2 were immersed in 300 mL of the prepared electrolyte solutions. Linear sweep voltammetry (LSV) was performed from 0 VRHE in the anodic direction at a scan rate of 10 mV/s, under both dark and illuminated conditions. The long-term stability of the PEC system was evaluated using CA at a constant potential of 1.23 VRHE. The applied bias photon-to-current efficiency (ABPE) result was calculated using the following equation.
Electrochemical impedance spectroscopy (EIS) was carried out in the frequency range from 10-3 to 106 Hz with an amplitude of 10 mV under a bias of 0.6 VRHE in the dark. Mott–Schottky (M–S) analysis was performed in the potential range from 0 to 1.23 VRHE at a frequency of 5000 Hz. The donor density (ND) of the photoanode was calculated from the M–S plot using the following equation:
where ε is the relative permittivity of BiVO4 (68), ε0 is the vacuum permittivity (8.85 × 10-12 F/m), and e is the elementary charge (1.602 × 10-19 C).
3. Results and Discussion
BiVO4 thin films were fabricated via a sol–gel-based spin coating process. Fig. 1(a) shows the variation in the film thickness as a function of the number of coating cycles. A linear increase in thickness was observed from two to five cycles; however, after the 6th cycle, further coating resulted in a significantly reduced thickness increment, indicating the onset of a saturation regime. This thickness behavior was clearly confirmed by the cross-sectional SEM images shown in the inset image, where the film thickness was measured to be 63.5 nm (2 cycles) and 167 nm (8 cycles). The minimal increase in thickness between the 6th and 8th cycles suggests that film growth reached a saturation point. This growth behavior is closely related to the surface morphology of the FTO substrate, as illustrated in Fig. 1(b). In the early stages of coating, the BiVO4 precursor solution preferentially filled the lower regions of the FTO substrate surface. During the 3rd to 5th coating cycles, uniform film formation continued the relatively planarized surface, resulting in a linear increase in thickness. However, once the surface is fully filled and the surface becomes flattened, further penetration of the precursor solution and nucleation are restricted, leading to a limited increase in the film thickness. This phenomenon corresponds to a saturation regime in the film-growth process, implying a practical limit to the achievable film thickness.
In Fig. 1(c), to investigate the effect of film thickness on PEC performance, LSV was conducted under AM 1.5G illumination in a 1.0 M KB electrolyte (pH 9.5). As shown in the results, when the film thickness doubled from two to four cycles, the photocurrent density increased by approximately 2.5 times. This improvement is attributed to enhanced light absorption and increased generation of photogenerated charge carriers as the film reaches sufficient thickness. However, when the coating exceeded four cycles, with an additional thickness increase of approximately 40 nm, the photocurrent density began to decline, with a similar decreasing trend observed after eight cycles. This behavior can be attributed to the inherently short diffusion length of minority carriers in BiVO4, which is typically on the order of several tens of nanometers. When the film becomes excessively thick, the photogenerated charge carriers are more likely to recombine within the bulk before reaching the surface, thereby reducing the charge transport efficiency and ultimately leading to a decrease in the photocurrent density. In contrast, the ultrathin BiVO4 film with two cycles exhibited significantly lower absorbance, as shown in Fig. 1(d), resulting in limited generation of photogenerated charge carriers and consequently insufficient photocurrent density. Therefore, the thickness of the BiVO4 film must be precisely optimized by balancing the light absorption and the charge transport distances. In this study, a film with a thickness of approximately 120 nm (corresponding to four coating cycles) demonstrated the highest photoelectrochemical performance.
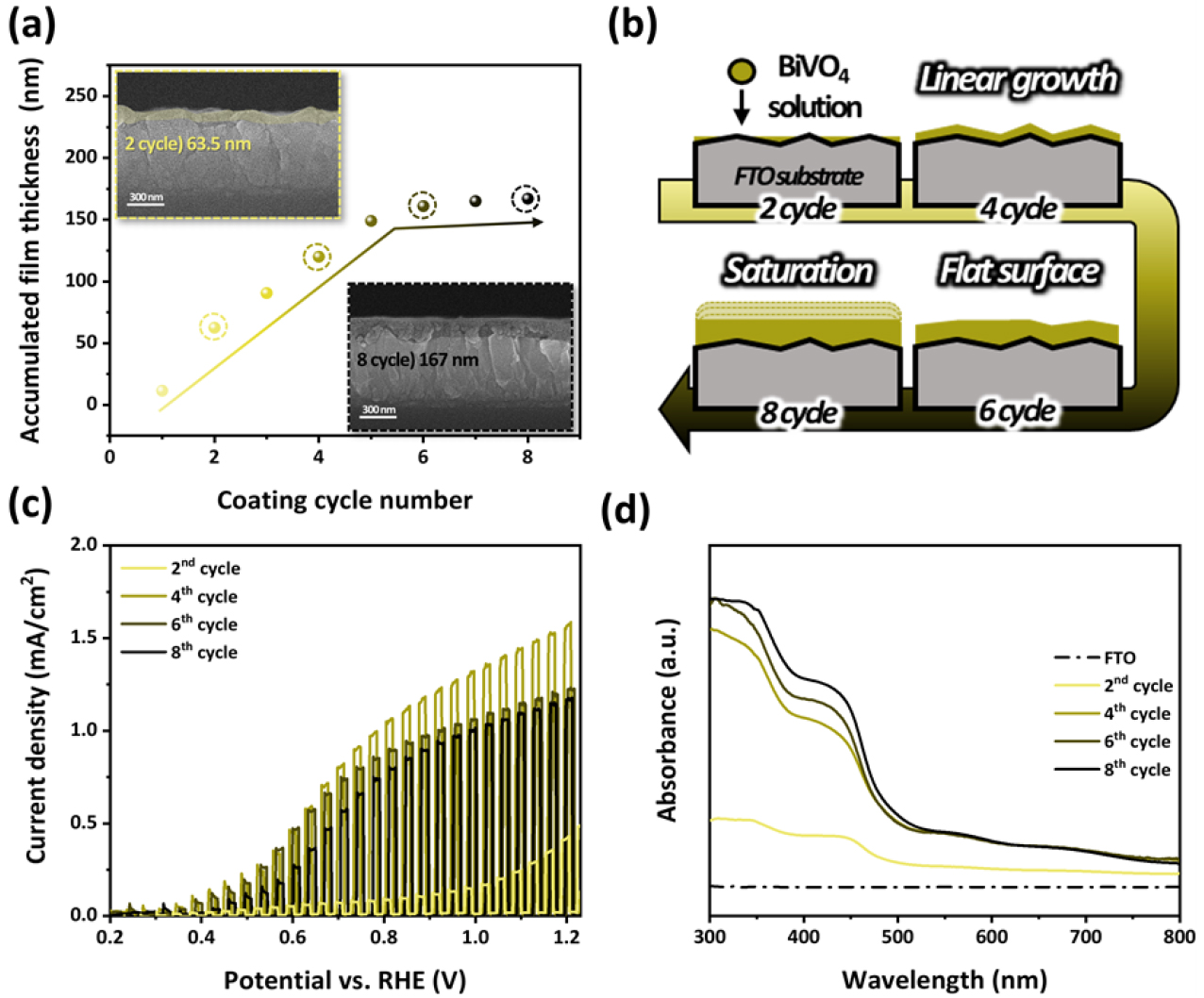
Fig. 1
(a) Thickness control of BiVO4 thin films as a function of spin-coating cycles and cross-sectional SEM images of BiVO4 films fabricated with 2 and 8 cycles. (b) Schematic illustration of film growth behavior (c) PEC performances of photoanodes in KB electrolyte, (d) UV-Vis absorption spectra of BiVO4 photoabsorber and FTO substrate
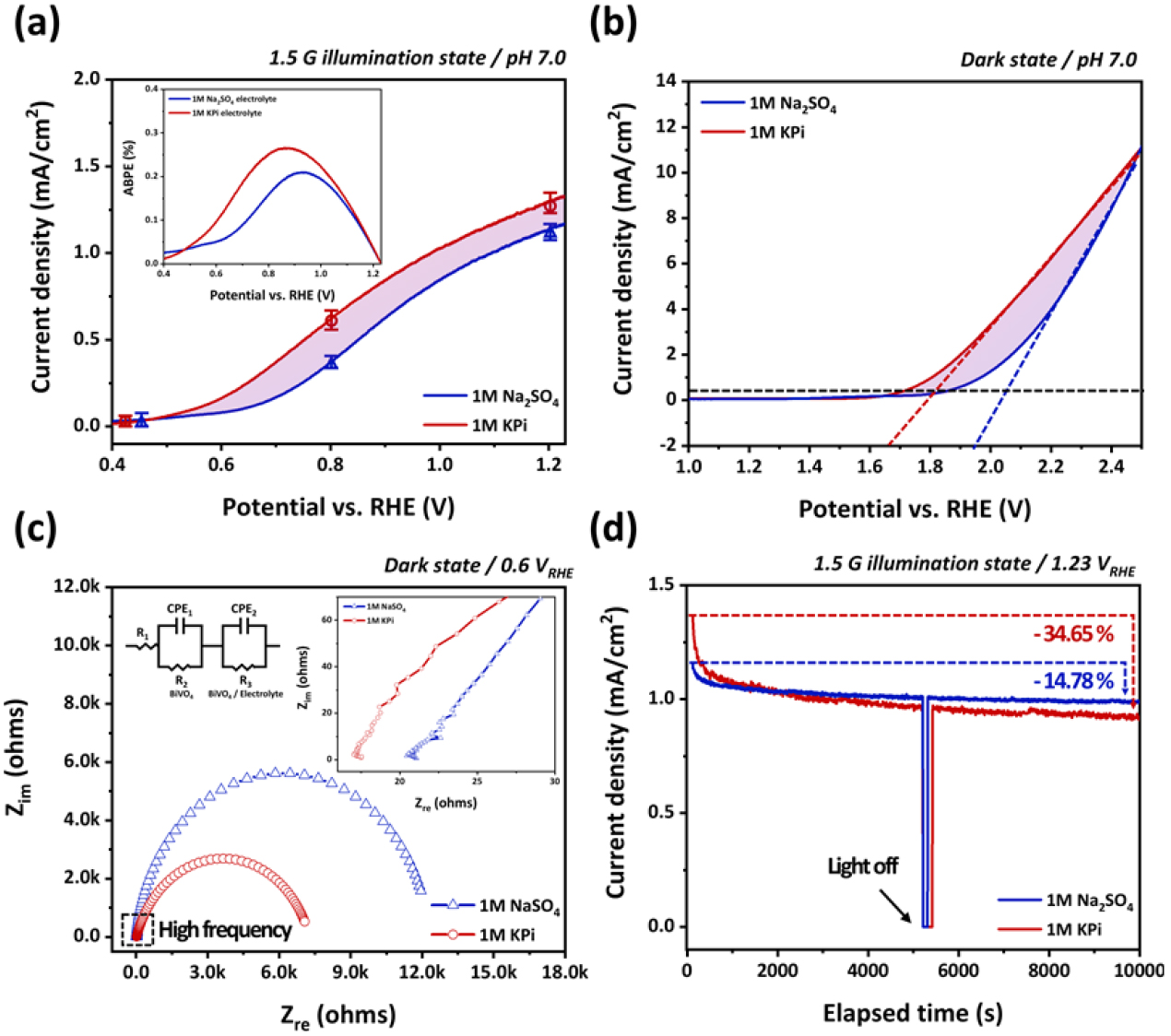
Nevertheless, to realize self-driven PEC hydrogen production in realistic seawater environments, it is essential to achieve stable OER operation under near-neutral conditions (approximately pH 6–8). To accurately evaluate the PEC behavior under neutral pH, we compared two widely used electrolytes, KPi buffer and Na2SO4 solution, both adjusted to an identical pH of 7.0, instead of the KB electrolyte, which exhibits instability owing to partial precipitation in the pH range of 6–7. As shown in Fig. 2(a), both electrolytes exhibited an OER at approximately 0.4 VRHE. At 1.23 VRHE, the BiVO4 photoanode in the KPi electrolyte exhibited a photocurrent density of 1.36 mA/cm2, whereas that in Na2SO4 was 1.21 mA/cm2. Notably, the KPi electrolyte consistently demonstrated a higher photocurrent over the entire potential range (0.4–1.23 VRHE). When interpreted alongside the ABPE results, this trend suggests that the KPi electrolyte enables a more efficient OER operation, even at lower overpotentials. These results were verified by repeated measurements of five independently prepared samples. As shown in Fig. 2(a), the error bars based on the mean values and standard deviations confirmed the quantitative reproducibility and reliability of the data for both electrolytes. In addition, the dark-state LSV measurements in Fig. 2(b) show a similar trend. The OER onset in the KPi electrolyte occurred at approximately 1.65 VRHE, whereas Na2SO4 required a higher potential, indicating the superior electrocatalytic activity of the KPi electrolyte.
Electrochemical impedance spectroscopy (EIS) analysis, presented in Fig. 2(c), quantitatively supported this conclusion. Measurements were conducted under dark conditions at a bias of 0.6 VRHE with an AC amplitude of 10 mV across a frequency range of 1 Hz to 1 MHz. The BiVO4 photoanode in the KPi electrolyte exhibited a smaller semicircle in the Nyquist plot, corresponding to a lower overall impedance and reduced charge-transfer resistance (Rct) compared to that in Na2SO4. Importantly, because the high-frequency region of the EIS spectrum reflects the intrinsic resistance of the electrolyte, the lower resistance observed for KPi further supports its superior electrochemical performance. Overall, these electrochemical analyses confirm that the KPi electrolyte provides a more favorable electrochemical environment for driving the OER compared to the Na2SO4 solution when interfaced with BiVO4 photoanodes. However, long-term stability testing revealed a trend contrary to that of the initial PEC performance. As shown in Fig. 2(d), after continuous PEC operation at 1.23 VRHE for more than 2 h, the photocurrent density in the Na2SO4 electrolyte decreased by approximately 14.78%, while in the KPi electrolyte, a more pronounced degradation of about 34.65% was observed. These results are consistent with previously reported literature, indicating that although KPi enhances the initial PEC activity, it suffers from reduced stability during prolonged operation owing to surface oxidation and electrode degradation. Therefore, to achieve both a high OER performance and long-term durability, an additional interfacial stabilization strategy is essential, particularly under PEC conditions involving KPi electrolytes. To address this trade-off between the OER performance and operational stability, a CoPi OEC was electrochemically deposited on the BiVO4 photoanode surface. CoPi not only provides active sites for the OER but also suppresses surface hole recombination. Moreover, phosphate ions (PO43-) present in the KPi electrolyte have been reported to facilitate and stabilize the formation of high-valent Co4+ intermediates through proton-coupled electron transfer (PCET) processes, thereby contributing to the sustained electrochemical reactivity of the catalyst layer. Such an electrolyte environment is thus considered to play a key role in maintaining both high OER activity and the structural stability of CoPi26-29.
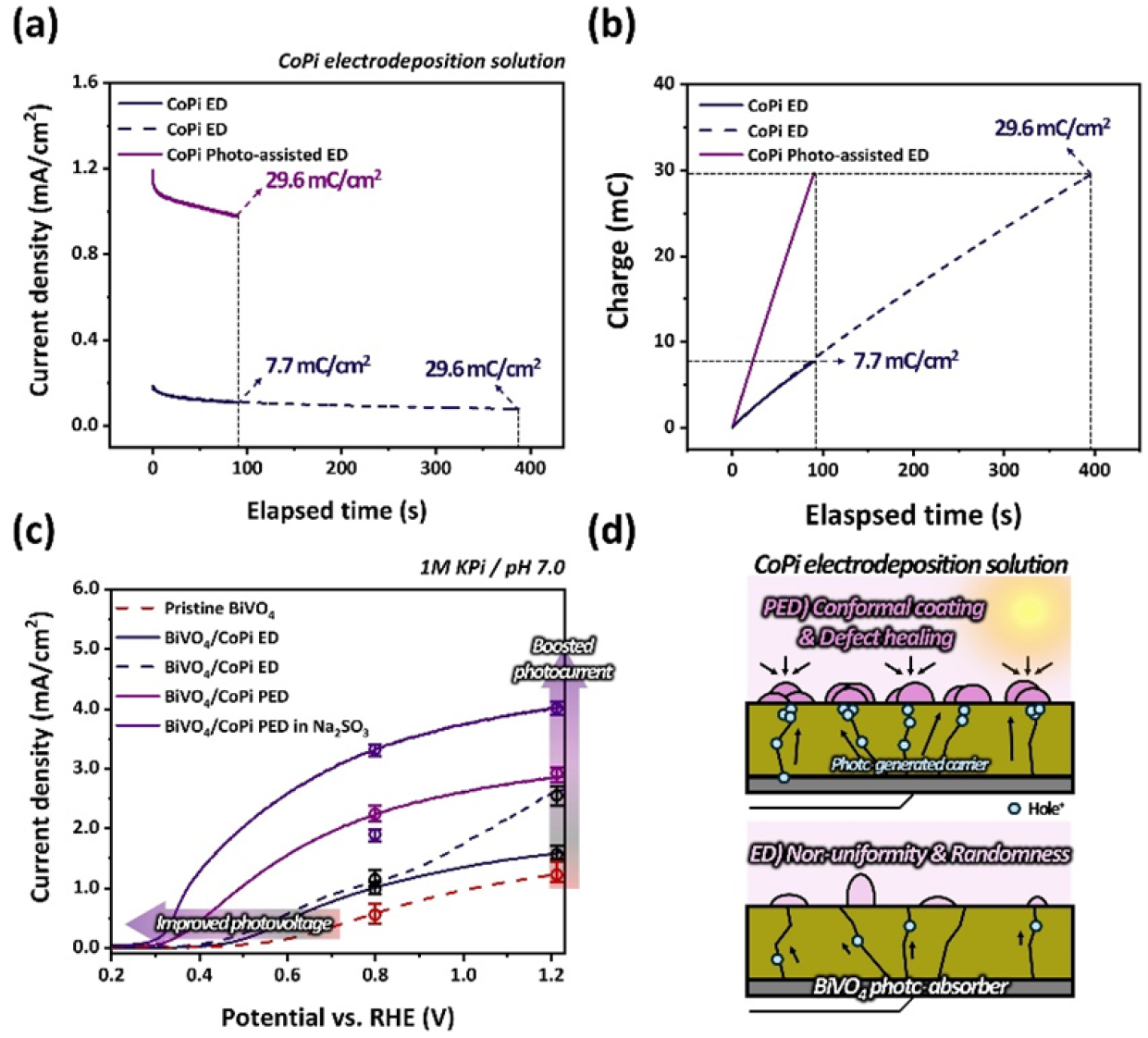
Traditionally, the CoPi OEC layer has been formed via conventional ED process under dark conditions. However, in this study, a PED technique was adopted as a more suitable approach for PEC reactions. Unlike ED, PED utilizes photogenerated holes produced under illumination, enabling site-selective CoPi deposition on photoactive sites. As shown in Fig. 3(a) and (b), to precisely control the nanoparticulate growth of CoPi, the deposition time was fixed at 90 s for both the conventional ED and PED processes. Under these conditions, the PED process accumulated a total charge of 29.6 mC/cm2, whereas the ED process yielded only 7.7 mC/cm2 within the same duration, clearly demonstrating a significant difference in current density and charge accumulation characteristics between the two methods. Accordingly, to rigorously verify that the enhanced performance of the PED process arises not merely from increased catalyst loading but from the intrinsic differences in the deposition mechanism, we conducted additional ED experiments under a matched total charge condition (29.6 mC/cm2). This required extending the ED deposition time by more than fourfold. The corresponding results are presented in Fig. 3(a) and (b).
As illustrated in Fig. 3(c), the BiVO4/CoPi (PED) electrode exhibited a photocurrent density of 2.89 mA/cm2, which is markedly higher than the 1.52 mA/cm2 observed for the BiVO4/CoPi (ED) electrode. Notably, the onset potential of the PED-deposited CoPi electrode was 0.26 VRHE, representing a cathodic shift of approximately 0.15 VRHE relative to pristine BiVO4, thereby clearly demonstrating simultaneous improvements in both photocurrent and photovoltage. In contrast, the ED sample prepared with the same total charge displayed comparable or slightly lower photocurrent density at 1.23 VRHE, but significantly inferior performance in the potential ranges from the onset to 1.1 VRHE. Furthermore, when Na2SO3 was added to the electrolyte as a hole scavenger, the BiVO4/CoPi (PED) electrode achieved a photocurrent density of approximately 3.98 mA/cm2, indicating that CoPi facilitated effective extraction of photogenerated holes and realized ~72% of the theoretical PEC efficiency of BiVO4. All measurements were repeated across five independently fabricated samples, showing consistent trends and low deviation. These results, as shown in Fig. 3(d), suggest that the prolonged ED process induces the formation of randomly oriented and non-uniform CoPi layers, which consequently degrade the interfacial quality between the catalyst and the photoelectrode. In contrast, the PED process maintains excellent PEC performance even under the same total charge condition. This can be attributed to the selective accumulation of photogenerated holes under illumination at surface defect sites such as oxygen vacancies or under-coordinated Bi/V atoms, which promotes uniform and localized CoPi growth. This hole-driven growth mechanism is consistent with previous studies indicating that charge carriers are selectively generated and utilized at illuminated photoactive sites30,31). Concurrently, it enables effective passivation of surface defects, thereby improving interfacial stability and charge separation efficiency, ultimately contributing to simultaneous enhancements in both photocurrent and photovoltage.
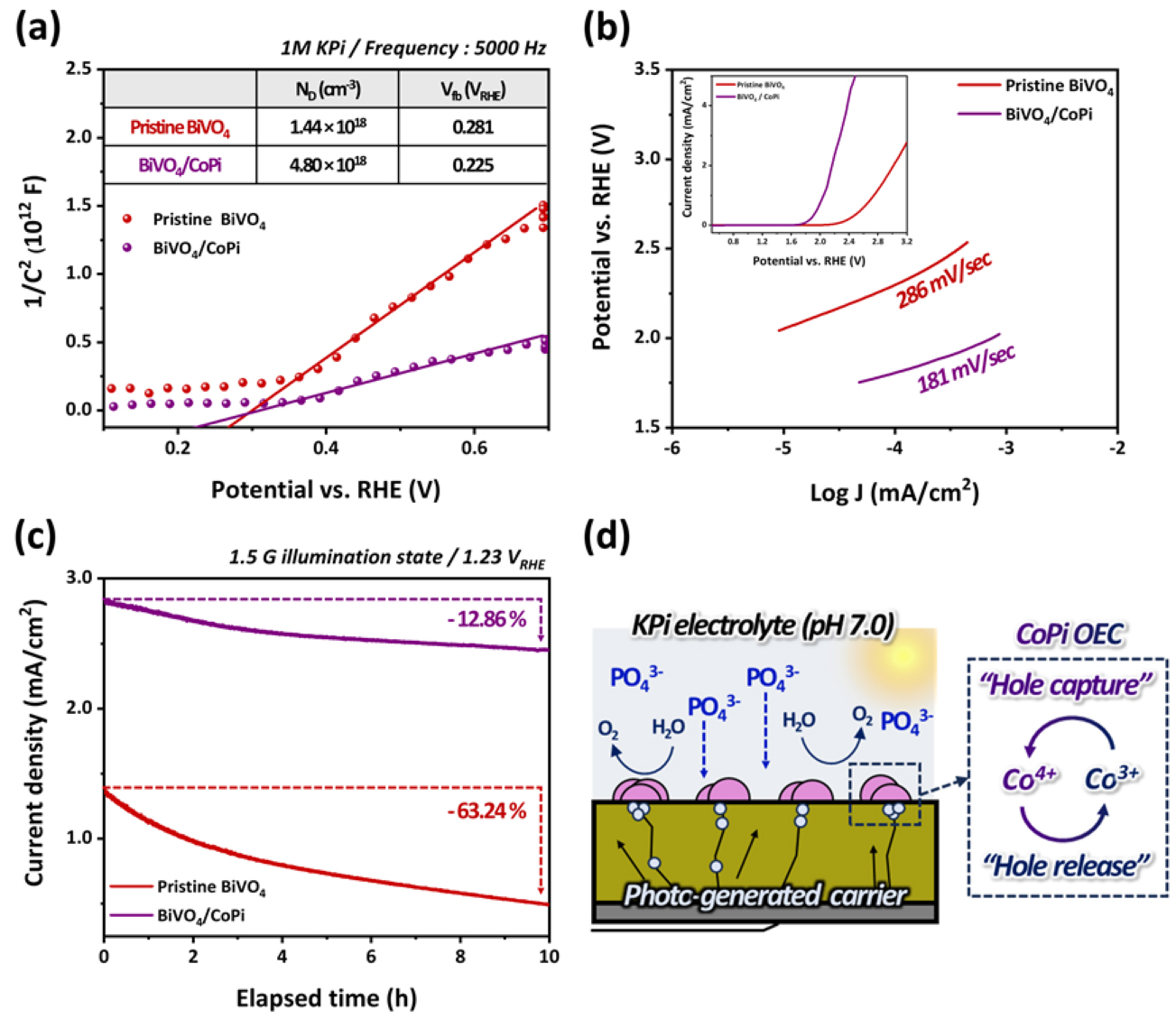
To evaluate the contribution of the CoPi OEC to the OER process quantitatively, a series of electrochemical analyses were performed. As shown in Fig. 4(a), the M–S analysis was conducted at 5000 Hz, where the space charge capacitance at the electrode–electrolyte interface dominates, based on the previously extracted equivalent circuit. The BiVO4/CoPi photoanode exhibited a flat-band potential of 0.225 VRHE, which was cathodically shifted from 0.281 VRHE for the pristine BiVO4. This shift indicates a more favorable energetic alignment with the OER reaction level. Additionally, the ND increased approximately fourfold, suggesting a significantly enhanced charge carrier concentration that facilitates hole transport to the surface under an applied bias. Fig. 4(b) shows the Tafel analysis, which further confirms the catalytic function of CoPi. The Tafel slope was reduced by approximately 1.5 times, demonstrating that CoPi effectively lowered the OER activation energy barrier and accelerated reaction kinetics. These improvements in the charge transfer and interfacial kinetics were also reflected in the long-term PEC stability results. In Fig. 4(c), after 10 hours of PEC operation at 1.23 VRHE, the pristine BiVO4 photoanode showed a photocurrent degradation of approximately 63.24%, whereas the CoPi-modified electrode exhibited only a 12.86% decrease, indicating significantly enhanced durability. As illustrated in Fig. 4(d), this improvement in stability is attributed to the stabilizing role of PO43– present in the KPi electrolyte, which helps to maintain the high-valent CoIV intermediate, thereby supporting sustained catalytic activity under prolonged OER conditions. The core mechanism of CoPi involves PCET associated with interconversion between the CoII/CoIII and CoIII/CoIV redox states. In this process, CoIV species serve as active sites for the OER, reacting with water molecules to evolve O2. The resulting Co species are then reduced to CoIII or CoII, completing a continuous catalytic cycle that maintains efficient oxygen evolution. As summarized in reactions (1)–(4), this reaction pathway indicates that CoPi facilitates the formation of surface intermediates such as –OH, O*, and OOH*, thereby accelerating OER kinetics. Simultaneously, the presence of PO43– ligands improves the proton transport pathways and effectively lowers the energy barrier under neutral or near-alkaline conditions. In Co-based OEC systems, photogenerated holes are initially captured by CoIII species (hole capture), which are subsequently oxidized to CoIV. These CoIV intermediates then participated in the oxygen evolution mechanism. After oxygen release, CoIV is reduced to CoIII, establishing a regenerative transfer loop that sustains the catalytic cycle (hole release)32).
Consequently, the CoPi cocatalyst serves as a key factor in fundamentally enhancing the charge-transfer efficiency in the PEC-driven OER by maximizing the utilization of photogenerated holes reaching the BiVO4 surface. Although CoPi has been widely employed as a cocatalyst to improve the electrochemical performance of BiVO4 photoanodes, its catalytic activity and long-term stability have shown limitations under certain electrolyte conditions, and few studies have systematically elucidated its operational mechanism in a structured framework. This study clearly identifies the role of CoPi based on synergistic interactions among the electrolyte, catalyst, and photoanode. In particular, the adoption of the PED strategy effectively maximizes the OEC performance of CoPi. These findings provide a strategic foundation for the practical realization of PEC-based green hydrogen production systems and offer a promising design principle for achieving high efficiency and durability under real-world operating conditions.
4. Conclusions
In this study, BiVO4 photoanodes with precisely controlled thicknesses were fabricated using a sol–gel-based thin-film process, and the effects of electrolyte conditions and cocatalyst deposition methods on the PEC water splitting performance were systematically investigated. By optimizing the trade-off between the light absorption and hole diffusion length, the spin-coating cycle was fine-tuned to achieve optimal PEC performance at a film thickness of approximately 120 nm. Comparative electrolyte analysis revealed that the KPi buffer exhibited superior OER activity compared with the Na2SO4 electrolyte, although its long-term operational stability was limited. To overcome this limitation, a CoPi cocatalyst was introduced to maximize efficiency under KPi conditions. Electrochemical analysis showed that CoPi enhanced PEC performance by improving hole transport and accelerating the OER kinetics on the BiVO4 surface through multivalent cobalt redox transitions and PCET mechanisms involving phosphate ligands. Notably, the PED technique enabled defect-passivated, uniform growth of CoPi nanoparticles, in contrast to the conventional ED processes. Consequently, the BiVO4/CoPi (PED) electrode exhibited an increased photocurrent density of 2.89 mA/cm2 and a cathodic shift in the onset potential by approximately 0.15 VRHE. Furthermore, even after over 10 h of continuous operation at 1.23 VRHE, the photocurrent decreased by only 12.86%, effectively overcoming the intrinsic stability limitations of pristine BiVO4. This study presents a strategic approach for precisely controlling the deposition of OER cocatalysts and elucidating the synergistic mechanisms among the electrolyte, catalyst, and photoanode. The findings offer practical insights for the design of PEC systems that satisfy both efficiency and durability requirements and lay a foundation for interfacial engineering aimed at stable solar hydrogen production. In addition, the PED method employed in this study can be readily performed under ambient conditions, is broadly applicable to various photoanode materials, and exhibits excellent scalability for large-area processing. These features highlight their strong potential for technology transfer to real-world PEC systems.